Day-Case Mastectomy: Artiss® (Fibrin Sealant Spray) « Contents
Razi, Kasra ST5 General Surgery KSS Deanery, East Sussex Healthcare NHS Trust
Cullen, Jack F2 East Sussex Healthcare NHS Trust
Arbis, Alex F1 East Sussex Healthcare NHS Trust
Shah, Elizabeth Breast Consultant, Conquest Hospital, East Sussex Healthcare NHS Trust
Corresponding author: Kasra Razi, 79 Redington Road, London NW3 7RR
Abstract
Aim: The use of drains in breast surgery is decreasing, and various methods of reducing the dead space after mastectomy have been described such as quilting of skin flaps and the use of adhesive tissue glues. The British Association of Day Surgery (BADS) outlines a 30% day-case target for simple mastectomy procedures. We conducted a feasibility study using a fibrin sealant spray (ARTISS) instead of drains in mastectomy without reconstruction, to reduce patient length of stay (LOS) & post-operative complications.
Method: A consecutive series of 50 patients, irrespective of age, BMI, social demographics & co-morbidities were included in the study. All surgical and theatre staff had appropriate training and data including day-case rates, seromas requiring aspiration and post-operative complications were prospectively collected.
Results: The mean age was 65 years; average BMI was 29.4kg/m2 and the average mastectomy weight was 712.4g. A day-case rate of 52% (26/50) was achieved. Of the day-case patients, 7 developed seromas requiring aspiration and one was re-admitted 7 days later with a haematoma. The overall seroma rate was 28% (14/50).
Conclusions: This study demonstrates that drain-free mastectomy is possible using ARTISS. The use of this product facilitates early discharge in all demographic groups and adherence to the national standard is achieved.
Introduction
Women undergoing a mastectomy for breast cancer have traditionally stayed overnight in hospital. Day-case surgery over the last decade has experienced an increase in practice with better outcomes for patients and improved cost-effectiveness for the NHS. The British Association of Day Surgery (BADS) advocates a 30% day-case target for simple mastectomy procedures (1), however overnight stay is still common practice. In our unit (East Sussex Foundation Trust) there has been a problem with establishing a day-case mastectomy programme for various reasons: our geographical location in the South East of England is home to one of the highest over 65’s population in the UK (2). An elderly patient demographic with multiple social issues can pose difficulties in initiating a day-case programme and the perceived complications associated with discharge to the community with drains in situ can increase hospital stay further. Our second stumbling block was a lack of nursing resource for drain removal. Nurses at our hospital did not have the physical space or time to remove drains in patients discharged home with drains in-situ, either in the community setting or as return ward attenders. The third hurdle was the mindset of patients and nursing staff towards early discharge of mastectomy patients for emotional and psychological reasons. Our patients had no confidence going home with their drain, as they were concerned about the drain-related pain. They were anxious about the drain not working or a complication forming or having a drain in-situ with children and family being around.
Seroma formation is the most common sequelae of breast surgery, with rates ranging between 15 to 81% of patients being affected (3). Seroma is the accumulation of clear serous fluid containing blood plasma and inflammatory fluid following surgery, however the pathogenesis and ways in which to reduce its formation are still poorly understood (4). The complications associated with seromas include infection, delayed wound healing, patient discomfort and skin flap necrosis, all of which can increase the likelihood of needing fluid aspiration of the seroma. Obliteration of the dead space between the wound surfaces following mastectomy appears to be fundamentally important in reducing seroma formation. Several methods have been proposed to prevent this; quilting of skin flaps, closed suction drainage and the application of adhesive tissue glues have all been described, however there is still no clear superior method (5).
The fibrin sealant spray Artiss® (Baxter, UK) is composed of a sealer protein solution with both human fibrinogen and human thrombin solutions. This allows for an extended polymerization time and full skin flap adherence to the wound bed. It is designed to reduce the formation of post-operative seroma and haematoma, thereby enabling a drain-less procedure (6). The literature evaluating the use of Artiss® in mastectomy is limited, however a series of studies have indicated a clear relationship between the use of Artiss® and the reduction of post-operative seroma (7,8). A recent study demonstrated a decrease in patient length of stay post procedure however the complication rate remained unchanged (9).
The purpose of this study was to analyse the efficacy of low-thrombin fibrin sealant (Artiss®) to reduce the length of hospital stay post mastectomy, and to additionally decrease the rate of seroma and haematoma formation.
Methods
Study Design
We designed a single centred unblinded prospective observational study looking at the effect of Artiss® on increasing day-case mastectomy rates. The trial was conducted between April 2018 and July 2019. All patients who required a simple mastectomy were given the option of using Artiss® instead of having the conventional Redivac drain. All patients who fitted the criteria of requiring a simple mastectomy regardless of breast size, body mass index (BMI) and co-morbidities were eligible. The primary aim was to increase the number of day-case mastectomies and reduce the hospital length of stay for all patients undergoing a simple mastectomy. The secondary aims were to reduce seroma and haematoma formation rates.
Prior to commencing the study all staff who made up the breast team including theatre nurses and breast care nurses were trained by a Baxter pharmaceutical company representative. A standardized protocol was used for the application of Artiss® in all patients. Only those trained in applying the product were able to use the sealant on a patient. (Protocol Inserted here)
Artiss® is a fibrosealant spray which has dual property which allows for tissue adherence and haemostasis. It is made up of two human plasma derived components. A sealer protein solution containing human fibrinogen and a synthetic fibrinolysis inhibitor that helps delay the premature breakdown of the clot at the site of application. A low-concentration human thrombin solution in a calcium chloride solution, which allows an extended polymerization time of 60 seconds compared to other fibrin sealants that can contain 500 IU/mL or more of thrombin. Each mastectomy flapped received 4mls of Artiss® spread evenly across the fascia covering pectoralis major. The mastectomy flap was then positioned neatly down on to the chest wall with 3 minutes of equal compression using a damp gauze. The skin edges were stitched using 3-0 vicryl and monocryl for the skin. All patients that had axillary lymph node dissection (ALND) had a size 10 Redivac drain placed in the axilla which was removed either when less than 50 ml output per 24 hours or after 5 days at the latest.
Postoperative Care
Patients were reviewed post surgery at the end of the day and assessed by the consultant before being discharged home. All patients were followed up in clinic two weeks after their operation by the consultant. At any time if patients felt that they may have developed a seroma or haematoma after they had been discharged they were seen by the breast enhanced recovery nurse and the breast consultant. If patients were symptomatic with either of these they were drained in clinic under an aseptic technique and the aspirated fluid was measured via the syringe. All measurements were recorded by the enhanced recovery after surgery (ERAS) nurse who was trained in aspirating mastectomy seromas.
Results
50 consecutive patients enrolled into this study. The mean (range) age of the patients was 65 (40-88) years with a mean (range) BMI of 29 (19-42) kg/m2. The average breast weighed 712g (143g to 2412g). 28 (56%) patients has invasive ductal carcinoma (IDC), 13 (26%) had invasive lobular carcinoma, 3 had ductal carcinoma in situ (DCIS), 1 lobular carcinoma in-situ (LCIS), 3 had risk reducing mastectomy and the remaining two had one had tubular carcinoma and the other papillary carcinoma. 25 patients (50%) had simple mastectomy with sentinel lymph node biopsy, 12 patients (24%) had simple mastectomy with ALND. All of those with ALND had a Redivac drain placed in the axilla which was removed when drain output was less than 50mls over 24 hours or at the very latest day 5 post operation. 26 patients (52%) went home the same day. 7 of those developed seromas (14%), 1 patient had their seroma drained twice. 21 out of the 24 patients (48%) who stayed longer stayed an extra day and 1 patient stayed 4 days. Of those who stayed longer, the majority of patients 15/24 (62.5%) stayed in longer due to patient choice, of those nearly half of them had a drain in their axilla. Other reasons for staying in longer included post-operative nausea and vomiting, blue dye reaction and cardiac complications. Overall, our hospital LOS for a simple mastectomy patients was reduced to a day-case procedure, previously 23-hour stay. On average the hospital LOS has reduced to half a day, prior to using Artiss® the average LOS was 1.5 days. Our day-case mastectomy rate has gone from 0% to nearly 50%. In total, 14 patients (28%) developed seromas requiring needle aspiration. 1 patient out of the fifty patients (2%) developed a haematoma which required evacuation in theatre. Our seroma rates have fallen from 40% previously audited by the department to 28%. It is too early to say whether Artiss® can reduce haematoma rate but the results so far as very promising.
Table 1 Demographic factors and clinical characteristics of all patients.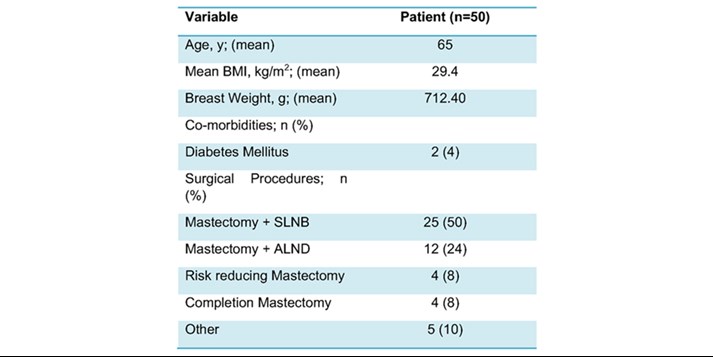
Discussion
Day-case rate
Our results have clearly demonstrated that Artiss® can help reduce hospital length of stay in patients who have a mastectomy. We have also managed to increase our day-case simple mastectomy rates from 0% to over 50%. By replacing the Redivac drain with Artiss® we have eliminated one of the main factors for delayed discharge. Having a drain in-situ and discharging patients with it can be anxiety inducing and often patients are reluctant to go home with a drain making it difficult for our unit to perform day-case mastectomies. The patient demographic in East Sussex varies compared to other parts of the country; the patients are generally older and for social reasons, some are simply unable to go home on the same day of procedure (1). Added to that the limited outpatient support for breast patients due to lack of resources makes it difficult for the clinicians to discharge the patient home safely with drains. Within the first year, our unit surpassed the 30% day case target from the British Association of Day Surgery (1). The introduction of this technique enabled a day-case mastectomy programme in our unit, which had previously faced challenges to achieving this. The cooperation between different members of the patient care pathway including surgeons, specialist breast nurses and enhanced recovery nurses supported the implementation of this change in practice.
Seroma and Haematoma Rate
Drains also pose other problems such as introducing infections into the wound, becoming blocked allowing for haematomas to form and cause patient discomfort. Other techniques have been used to mimic the effects of the drain without needing to worry about the issues that come with them such as quilting but with the properties of Artiss® we thought we could tackle other issues associated with mastectomies such as seroma and haematoma formation. The haemostatic properties of Artiss® speed up the process of coagulation, which reduce the likelihood of haematoma formation. The human fibrinogen component can enable lymphatic vessel closure, hence reducing postoperative dead space and seroma formation through tissue adhesion (4). Although we only had one haematoma however with the small number of patients, we have it is too early to say whether Artiss® has led to any improvement.
Thirteen patients had drains placed in their axilla following ALND. Only two of these patients went home the same day and on average stayed one day longer than those without. It was the patient’s choice to stay in a day longer, none of these patients felt comfortable going home with a drain. This clearly demonstrates the hindrance drains have on performing day-case mastectomy. Seroma formation is a common complication of breast mastectomies. The current literature quotes incidence of up to 90%. Various studies which have investigated this have identified that seroma incidence rates can be reduced by flap fixation (Kuroi K et al).
Before introducing Artiss® the department seroma rates were around 40% this has now reduced to 26%. When analyzing the data on those who developed seromas there is no clear reason for this; they were not necessarily older or bigger breasted. The mean (range) weight of those who developed seromas was 636.7mg (385-866mg) below that of the cohort average. Of those that developed seromas and went home the same day 5 had simple mastectomy and SLNB the others had ALND instead. The mean BMI was also 27.2 kg/m2 in seroma cohort, 2 units below the entire group mean BMI.
The aspiration of seromas from the surgical wound is a significant cause of patient discomfort, can delay further treatment, increase the rate of wound infections and lead to repeated outpatient visits (3). The importance of reducing seroma formation is clinically relevant for our cohort of patients – the introduction of Artiss® has slightly reduced the frequency of seromas however their incidence remains significant.
Remission Rates
We had a 0% remission from our cohort of patients.
Limitations
The main limitation of this study is that it is a prospective observational study with no control group. No statistical analysis of the data has been performed and the lack of a control group and group randomisation would question the validity of the results. The trial is also at risk of the Hawthorne effect since both the clinician and patient were aware that they were receiving a new treatment to increase day case mastectomy rates. Behaviours and decisions from both patient and clinician may be influenced therefore questioning the validity of the results of this study. To improve our study and assess the validity of the result further trial in the form of randomised control trials (RCTs) are needed.
Conclusion
It is feasible to use Artiss® instead of drains to allow earlier discharge of patients and make day-case mastectomy a reality. The product can also helped reduce both the number and the size of the seroma in mastectomy patients by obliterating the dead space. To develop this project further a trial looking at cost-analysis and patient satisfaction is required to help us decide whether there is any place for drains in patients with a simple mastectomy.
References
- Skues M. BADS directory of procedures (4th edn). London: British Association of Day Surgery; 2012.
- Office for National Statistics (ONS) 2017: https://www.ons.gov.uk/peoplepopulationandcommunity/populationandmigration/populationestimates/articles/overviewoftheukpopulation/july2017
- Pogson CJ, et al. Seroma following breast cancer surgery. Eur J Surg Oncol. 2003; 29:711–717. doi: 10.1016/S0748-7983(03)00096-9.
- R. Benevento, A. Santoriello, G. Pellino, G. Sciaudone, G. Candilio, G.S. De Fatico, F. Selvaggi, E. Colo, S. Canonico. The effects of low-thrombin fibrin sealant on wound serous drainage, seroma formation and length of postoperative stay in patients undergoing axillary node dissection for breast cancer. A randomized controlled trial. Int. J. Surg., 12 (2014), pp. 1210-1215
- van Bastelaar, J., Beckers, A., Snoeijs, M. et al.Flap fixation reduces seroma in patients undergoing mastectomy: a significant implication for clinical practice. World J Surg Onc14, 66 (2016) doi:10.1186/s12957-016-0830-8
- van Bastelaar, J., Theunissen, L.L.B., Snoeijs, M.G.J., Beets, G.L. & Vissers, Y.L.J. 2017, Flap Fixation Using Tissue Glue or Sutures Appears to Reduce Seroma Aspiration After Mastectomy for Breast Cancer.
- Sakkary MA. The value of mastectomy flap fixation in reducing fluid drainage and seroma formation in breast cancer patients. World J Surg Oncol. 2012;10.
- Ten Wolde B, Van Den Wildenberg F, Keemers‐Gels ME, Polat F, Strobbe L. Quilting prevents seroma formation following breast cancer surgery: closing the dead space by quilting prevents seroma following axillary lymph node dissection and mastectomy. Ann Surg Oncol. 2014; 21:802‐807.
- Harrison C, Remoundos DD, Harvey KL, Stoker GV, MacLean G, Adwani A et al (2019) Increasing the incidence of drain-free day-case mastectomies with the use of a fibrin tissue sealant; data from a single surgical center in the United Kingdom. Breast J 25(6):1090–1096
Cite this article as https://daysurgeryuk.net/en/resources/journal-of-one-day-surgery/?u=/2020-journal/jods-303-august-2020/day-case-mastectomy-artiss-fibrin-sealant-spray/
Download this article as PDF here: https://appconnect.daysurgeryuk.net/media/42064/razi_-day-case-mastectomy.pdf